 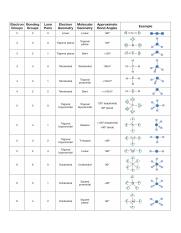 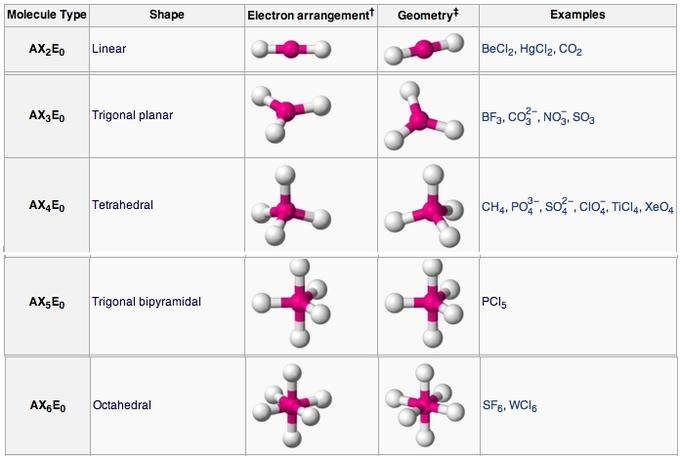
These processes are discussed extensively in your CHEM-C 105 textbook and lecture notes, and should be reviewed before you attend the laboratory session. The flow chart below gives a schematic view for how one can figure out the molecular geometry from the molecular formula: From molecular formula to molecular geometry and polarity. The molecular geometry of a species affects crystal structure, phase change behavior, energy and rates of chemical interactions, solubility, and virtually every other characteristic of that compound.īefore beginning to study the geometries of covalently-bonded groups, it is necessary to have a clear picture of which atoms are present and which atoms are bonded directly together in the grouping. The molecular geometry has a direct impact on the compound’s physical properties and chemical reactivity. The bonding pattern is the Lewis structure. Depending on the bonding pattern about each of the different central atoms in a molecule, each molecule will have a different 3D structure – the molecular geometry. Ĭovalent bonds are formed when electrons are shared between the atoms in a molecule. In this experiment, we focus on molecular compounds, which are held together by covalent bonds. There are different types of chemical bonding involved in making compounds. In reality, we have many different types of compounds, each with distinct structures. The paucity of materials with which we can live with based on elements alone is beyond imagination. We have 92 naturally occurring (plus a number of artificially made) elements, and that’s it. Life would be very boring if we tried to live with elements alone. Tro, Chemistry: Structures and Properties, Ch.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
